Clementine
Well-Known Member
I agree with what Bobby says here I was discussing why bottled conditioned beer takes longer to than just the time for the yeast to consume the sugar to be properly carbonated and have nice fine bubbles. Everyone seems to think that it was the CO2 dissolving into the beer from the head space, my opinion is that as the beer is allowed to settle and the yeast and sediment in the beer drop from solution that their is less nucleation sites for the CO2 to come out of solution and hence a finer bubble. The CO2 is excreted by the yeast into solution one molecule at a time the excess dissolved beer is off gassed into the head space.
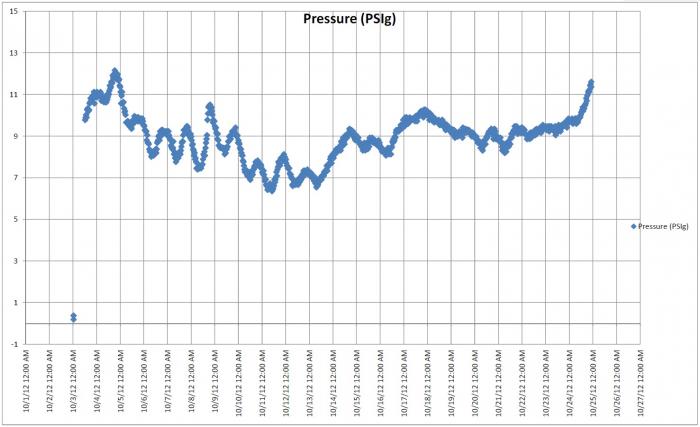
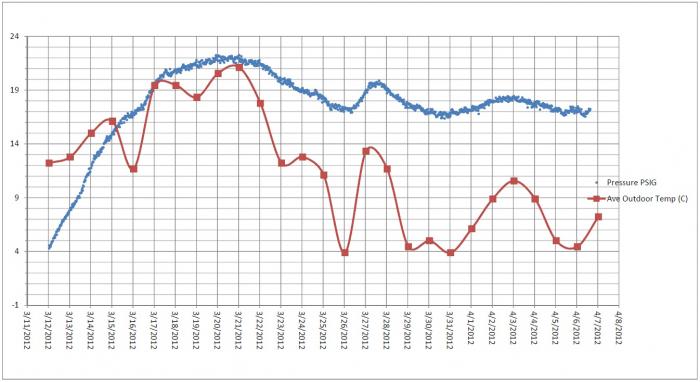
CO2 in the head space of a carboy vs CO2 in the beer is extremely complicated when you look at the law of partial pressures and the affect of gasses exchanging through the water/alcohol in the air lock and the back pressure of gasses caused by the airlock it is enough to make you go crazy. This effects your carbonation level as the dissolved CO2 concentration is added to your total CO2 in most priming calculators.
If you ever want to see the law of partial pressures in action get one of those stupid pump caps that someone thought would allow you to keep soda carbonated by pumping atmospheric air into the head space of a opened soda bottle.
http://www.amazon.com/dp/B00004XSH3/?tag=skimlinks_replacement-20
It does not take into account that air is only 1% CO2, when I saw those in the super market I thought that was so funny, it was a tax for those people who were not listening in chemistry class.
Head space to beer ratio matters, I think the less head space the better as then you have less O2 in the beer during the bottling process but back off your sugar to get the desired results. All the pressure in the bottle is equal through out liquid and gas, if your head space is the same volume (determined by the bottle wand) and you are producing the same amount of CO2 your pressure will be higher.
Think of it like this 12oz bottle = 12 oz of beer with a set amount of sugar produces X volumes of CO2. 24oz of beer with the same concentration of priming sugar with produce 2X volumes of CO2. Now the head space has stayed the same so the ratio has been increased resulting in higher pressure. It is a lot more complicated than that as you have to throw in there how much space the water (beer) molecules take up so that will further skew the pressure relationship, throw in temp changes effecting the CO2 solution and it is really complicated. I don't know of a priming calculator for different size bottles but I would start with backing off the sugar concentration by 5 to 10% to start with and see how it goes. I enjoy my beer on the lower side of the carbonation style so I would rather under shoot than over shoot.
Clem
CO2 in the head space of a carboy vs CO2 in the beer is extremely complicated when you look at the law of partial pressures and the affect of gasses exchanging through the water/alcohol in the air lock and the back pressure of gasses caused by the airlock it is enough to make you go crazy. This effects your carbonation level as the dissolved CO2 concentration is added to your total CO2 in most priming calculators.
If you ever want to see the law of partial pressures in action get one of those stupid pump caps that someone thought would allow you to keep soda carbonated by pumping atmospheric air into the head space of a opened soda bottle.
http://www.amazon.com/dp/B00004XSH3/?tag=skimlinks_replacement-20
It does not take into account that air is only 1% CO2, when I saw those in the super market I thought that was so funny, it was a tax for those people who were not listening in chemistry class.
Ever since I switched to 1/2 liter swing-top bottles, I've been having trouble with over carbonation and gushering, even after 2months in the bottle.I'm beginning to think I'm not leaving enough head space. With 12 oz. bottles, I would just use the displacement of the bottling wand to set the correct headspace. Does that work with larger bottles or should I leave a bit more?
Head space to beer ratio matters, I think the less head space the better as then you have less O2 in the beer during the bottling process but back off your sugar to get the desired results. All the pressure in the bottle is equal through out liquid and gas, if your head space is the same volume (determined by the bottle wand) and you are producing the same amount of CO2 your pressure will be higher.
Think of it like this 12oz bottle = 12 oz of beer with a set amount of sugar produces X volumes of CO2. 24oz of beer with the same concentration of priming sugar with produce 2X volumes of CO2. Now the head space has stayed the same so the ratio has been increased resulting in higher pressure. It is a lot more complicated than that as you have to throw in there how much space the water (beer) molecules take up so that will further skew the pressure relationship, throw in temp changes effecting the CO2 solution and it is really complicated. I don't know of a priming calculator for different size bottles but I would start with backing off the sugar concentration by 5 to 10% to start with and see how it goes. I enjoy my beer on the lower side of the carbonation style so I would rather under shoot than over shoot.
Clem
Last edited by a moderator: