CodeSection
Well-Known Member
- Joined
- Feb 4, 2018
- Messages
- 1,655
- Reaction score
- 818
I think others may benefit from what I have been experiencing. I have been using MME for sometime and found it very useful in my brews. I like the layout and the recommendations. The results are spot on and it is my goto program.
With that said, last week I was changing a recipe so I decided to compare MME's recommendation to that of BS3's and BF's recommendations on a brew (which I just happened to finished at 4:45AM this morning, btw). To my surprise, there were huge differences...so far off that I thought I had made a mistake somewhere.
MME 6.70 version, showed I needed to add 6g of baking soda to reach my target mash pH of 5.40.
When I entered the same mineral additions into BeerSmith3 I got an estimated pH of 5.59 and it was suggested to add 12.0 ml of lactic acid.
When I entered the information in Brewer's Friend, I got an estimated pH of 5.58 and it was suggested to add 8.96 ml of lactic acid.
When I removed the 6g of baking soda, MME shows pH at 5.28 (would need add 6g baking soda to get to 5.40), BS3 5.49 (would need to add 5.7ml lactic acid) , and BF 5.48 (would need to add 3.42 ml lactic acid).
With the discrepancies above I thought I was doing something wrong. So, I reached out to the developer of MME asking for help. He believed his program was accurate as I was using a lot of caramel malt 90L which is more acidic. I went with MME and added the 6g of baking soda it recommended. I am sure glad I did!
This batch was totally different in mashing. Rather than a single infusion, I had four mash steps. Instead of mashing for an hour, I mashed for 80 minutes. I used 6.00g of baking soda.
pH @ 30 minutes @ 68F: 5.55
pH @ end of mash @ 68F: 5.43
pH @ post boil @ 68F: 5.31
I'm happy with the results, especially since I changed the recipe and my process.
Had I used any of the other two programs, I would have been way off since they were suggesting adding a large amount of lactic acid.
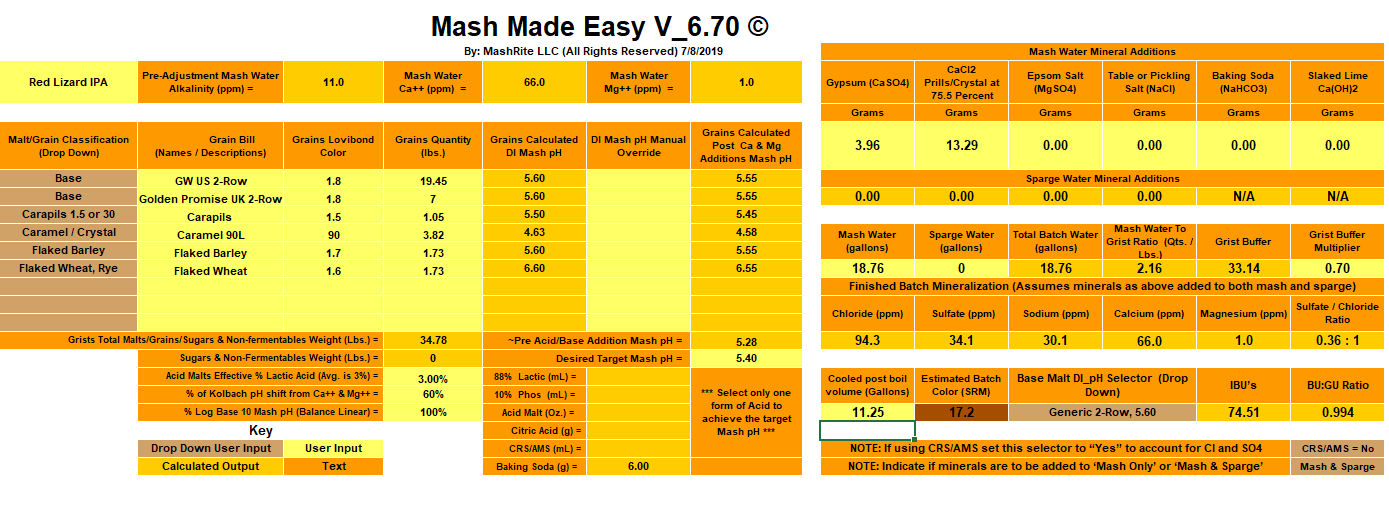
Below is a screen shot of my MME recipe...
I highly recommend you download and check out Mash Made Easy. I think you may be surprised just how easy and accurate it is. This is not a paid endorsement and I do not know the developer.
With that said, last week I was changing a recipe so I decided to compare MME's recommendation to that of BS3's and BF's recommendations on a brew (which I just happened to finished at 4:45AM this morning, btw). To my surprise, there were huge differences...so far off that I thought I had made a mistake somewhere.
MME 6.70 version, showed I needed to add 6g of baking soda to reach my target mash pH of 5.40.
When I entered the same mineral additions into BeerSmith3 I got an estimated pH of 5.59 and it was suggested to add 12.0 ml of lactic acid.
When I entered the information in Brewer's Friend, I got an estimated pH of 5.58 and it was suggested to add 8.96 ml of lactic acid.
When I removed the 6g of baking soda, MME shows pH at 5.28 (would need add 6g baking soda to get to 5.40), BS3 5.49 (would need to add 5.7ml lactic acid) , and BF 5.48 (would need to add 3.42 ml lactic acid).
With the discrepancies above I thought I was doing something wrong. So, I reached out to the developer of MME asking for help. He believed his program was accurate as I was using a lot of caramel malt 90L which is more acidic. I went with MME and added the 6g of baking soda it recommended. I am sure glad I did!
This batch was totally different in mashing. Rather than a single infusion, I had four mash steps. Instead of mashing for an hour, I mashed for 80 minutes. I used 6.00g of baking soda.
pH @ 30 minutes @ 68F: 5.55
pH @ end of mash @ 68F: 5.43
pH @ post boil @ 68F: 5.31
I'm happy with the results, especially since I changed the recipe and my process.
Had I used any of the other two programs, I would have been way off since they were suggesting adding a large amount of lactic acid.
Below is a screen shot of my MME recipe...

I highly recommend you download and check out Mash Made Easy. I think you may be surprised just how easy and accurate it is. This is not a paid endorsement and I do not know the developer.