hopjuice_71
Well-Known Member
- Joined
- Mar 26, 2016
- Messages
- 373
- Reaction score
- 374
Edit: This one threw me a curve ball I can't hit (as in comprehend):
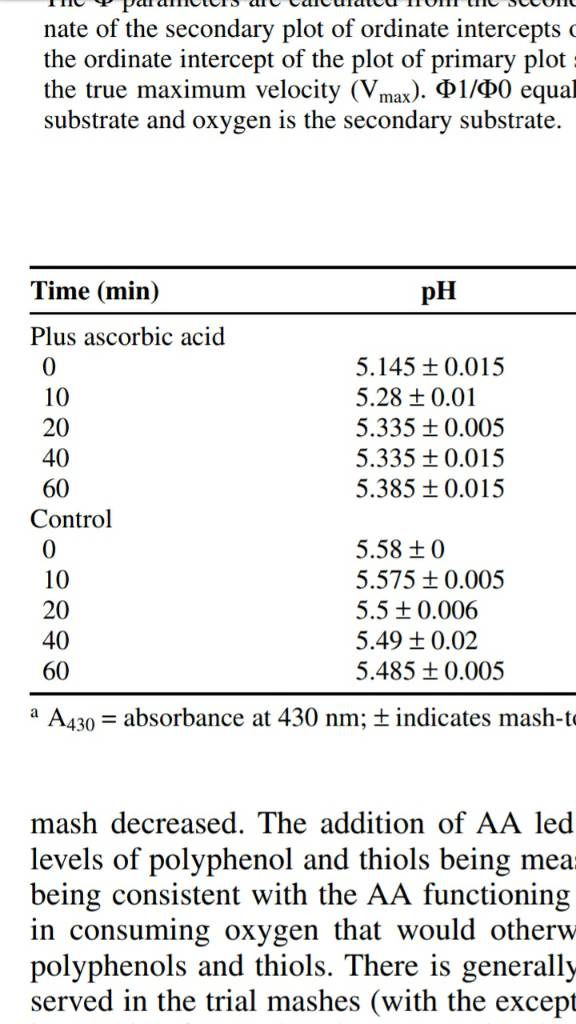
If it says anything to me, it's saying that alpha acids are better solubilized at higher pH's, but once you get them to fully solublize in a low pH environment nothing else will be changed by the lower pH aside from the initial solubilization time. And isomerization must be preceded by solubilization. Can that be what it is saying? Again I must inject that pellet hops solubilize leagues faster than whole/leaf. Are pellets the answer to the negation of slower solubilization due to lower Wort pH?
It was not well articulated, but this is what it means. The isomerization reaction obeys first-order kinetics, which means that the higher the concentration of a-acids, the higher the velocity of the reaction to iso-a-acids. So, higher solubility at low pH means more a-acid, which means more production of iso-a-acids. That is the first part of the sentence. The last part of the sentence reflects that there is no inherent change to the kinetic constant governing the reaction at different pHs. Practically speaking, it seems like a bit of a strange sentence as I believe you would have to get to quite extreme hopping levels to reach solubility limits at boiling temperature, so pH really shouldn't feature much there (maybe possible in the days of the IBU race?).
Last edited: