Obese Chess
Well-Known Member
Good afternoon!
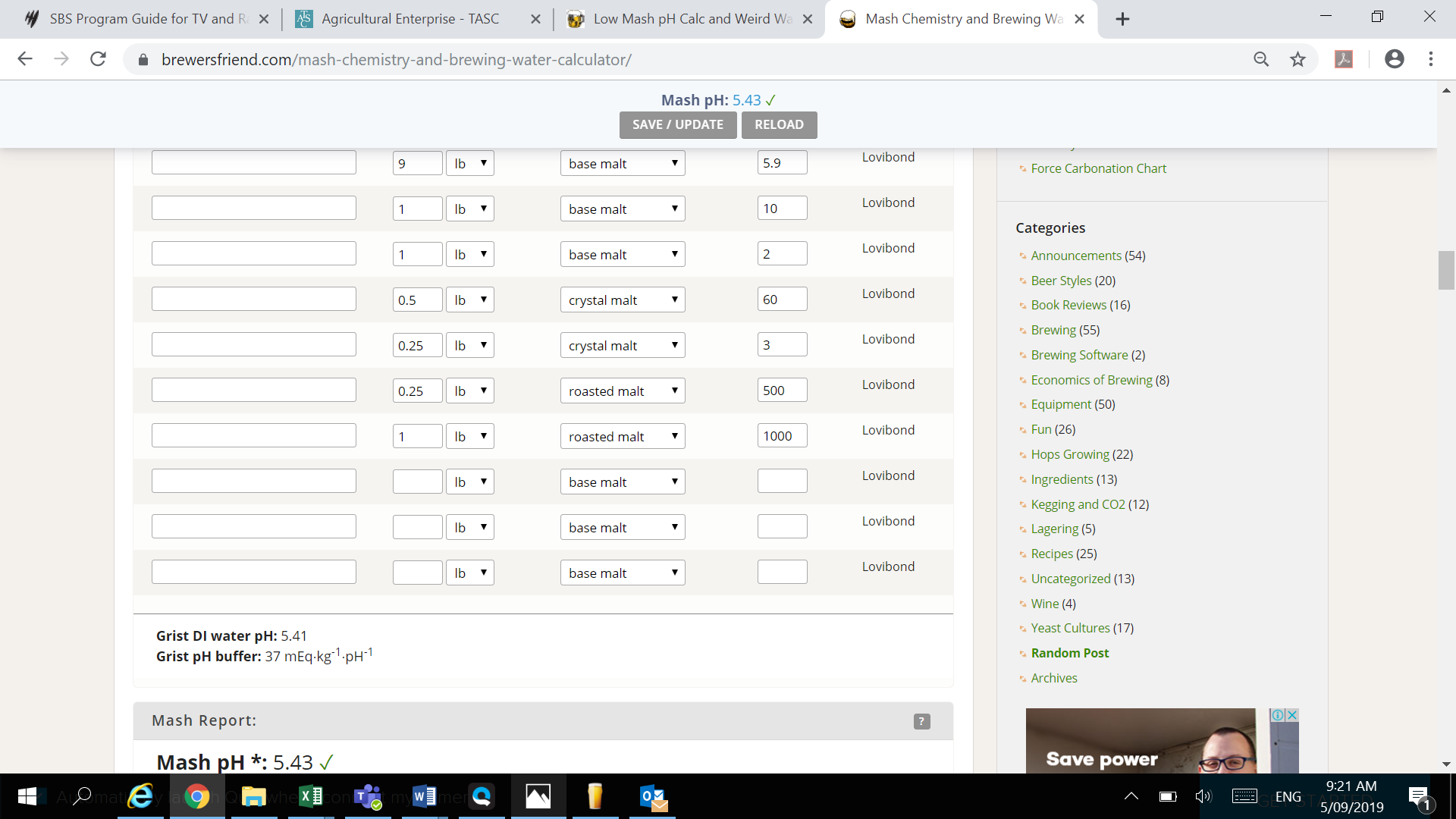
I am a few days from my first ever brew day and trying to get the last bits of information dialed in on a Robust Porter whose recipe I keep changing from its original (lol). I plugged my grain bill into Brewers Friend and was given a potential mash pH of 4.1 without inputting any sort of water profile. Realizing this to be impossible, especially because removing 3/4lbs of chocolate malt from my 13lb grain bill bumped me back up to 5.8, I could have just ignored it - but instead I started messing around with water chemistry, despite multiple local homebrewers, the guy who originally wrote the recipe, and my LHBS telling me that our water here is just fine and not to mess with it (I still may do this, but I've got time to kill).
I got the following water profile from my supplier:
I did some more digging and found other water profiles in my area putting our sulfate at 5 ppm, so I'm using that as my value. For everything else I'm just taking the midpoint in those ranges (24ppm for Ca+2 and Mg+2 and 26.5ppm for HCO3-). When I plug this into Brewers Friend, my mash pH goes up to a whopping 4.18. In Bru'N Water, these inputs show up as being "unbalanced" but I can't figure out why.
I imagine my "averaging" - or "averaging" on the part of the utility company - is what's throwing these numbers off. What am I doing wrong and how can I fix it? For what it's worth, if I plug this same recipe into Beersmith I get a mash pH of 5.48 or something around there.
Thanks!
I am a few days from my first ever brew day and trying to get the last bits of information dialed in on a Robust Porter whose recipe I keep changing from its original (lol). I plugged my grain bill into Brewers Friend and was given a potential mash pH of 4.1 without inputting any sort of water profile. Realizing this to be impossible, especially because removing 3/4lbs of chocolate malt from my 13lb grain bill bumped me back up to 5.8, I could have just ignored it - but instead I started messing around with water chemistry, despite multiple local homebrewers, the guy who originally wrote the recipe, and my LHBS telling me that our water here is just fine and not to mess with it (I still may do this, but I've got time to kill).
I got the following water profile from my supplier:
Ca+2 and Mg+2: Calcium and Magnesium analysis determines water hardness. Our water is very soft, with hardness ranging from about 18 to 30 ppm.
Na+:11 ppm.
Cl-: 2 ppm.
SO4-2 is sulfate and is not something that we analyze for.
HCO3-: Bicarbonate analysis determines alkalinity. Our water ranges from about 23 to 30 ppm.
pH runs about 7.7.
I did some more digging and found other water profiles in my area putting our sulfate at 5 ppm, so I'm using that as my value. For everything else I'm just taking the midpoint in those ranges (24ppm for Ca+2 and Mg+2 and 26.5ppm for HCO3-). When I plug this into Brewers Friend, my mash pH goes up to a whopping 4.18. In Bru'N Water, these inputs show up as being "unbalanced" but I can't figure out why.
I imagine my "averaging" - or "averaging" on the part of the utility company - is what's throwing these numbers off. What am I doing wrong and how can I fix it? For what it's worth, if I plug this same recipe into Beersmith I get a mash pH of 5.48 or something around there.
Thanks!